Microscopy & Microtechniques
Micropatterning: The art of micro-controlling cells
Nov 06 2020
Author: Marie-Charlotte Manus, Dr Louise Bonnemay, Matthieu Opitz on behalf of Alvéole
The challenge of studying living cells in vitro
Like all living beings, cells are sensitive to their environment which influences their behaviour and development (cell differentiation, proliferation, migration, etc.). Indeed, several studies have shown that mechanisms such as cancerous growth or stem cell differentiation are not only induced by genetics but also by the cellular environment [1,2]. However, its complexity in terms of biochemistry, structure or interactions explains the difficulties faced when studying living cells in vitro. In addition to this problem of physiological relevance, researchers are faced with many other challenges, such as the recurring reproducibility issues, ease of use and efficiency, even more true in the case of high throughput.
Among the experimental tools and techniques used to solve these issues is micropatterning: a method routinely used in mechanobiology, known as the in vitro analysis of living cells intra and inter-cellular mechanisms. But far from being limited to mechanobiology, micropatterning can be a powerful asset for different fields of research, such as disease modelling, immunology, toxicology. It also recently appeared as a must-have for the promising field of cryo-electron tomography (cryo-ET): a technique based on cryo-electron microscopy (cryo-EM) which allows the imaging of macromolecular complexes in flash frozen cells and therefore a better understanding of their role in their native state.
This article will investigate the benefits of micropatterning in cell biology studies and then for the emerging field of whole-cell cryo-ET.
Micropatterning is defined as the controlled deposition of biomolecules at the micron scale aiming to mimic or control the cell biochemical microenvironment. It was first used in the cells biology research about 30 years ago to investigate the link between cell shape (using adhesive proteins to locally control cell adhesion and shape) and terminal differentiation [3]. Since then, micropatterning has been routinely used to demonstrate the link between cell adhesion, cell shape and intra-cellular organisation [4,5].
Different micropatterning techniques have been developed and improved over the years, first by academic research labs and then by industrial companies to broaden its access, facilitate its use and guarantee reproducible results. Nowadays, the two most commonly used micropatterning approaches are microcontact printing using elastomer stamps and deep UV photolithography through photomasks [6]. These approaches however lack some capabilities now often considered mandatory, including flexibility in pattern shapes, multi-substrates compatibility (due to a growing use of soft or micro structured substrates) and alignment on these substrates; these limitations constrained the use of micropatterning to plain flat substrates for a long time.
Furthermore, the increased complexity of cell biology experiments caused an increased diversity of substrates with an increased fragility, due to their material composition, low stiffness or microscopic size. A typical example of that fragility would be the electron microscopy grids (EM grids) used for whole-cell cryo-electron tomography [7] - a circular substrate about 3 mm diameter, composed of a metallic mesh grid and a carbon layer film approximately 20 to 50 nm thick.
Recently, one micropatterning solution seemed to answer all these issues of design flexibility, alignment and surface preservation by preventing direct contact with the substrate. This innovative system is the PRIMO® maskless photopatterning system developed by Alvéole, a French spin-off from Centre National de la Recherche Scientifique, France (CNRS), funded in 2010, the mechanism of which will be presented in this article among others.
Micropatterning workflow: A combination of repulsion and attraction
All protein micropatterning techniques rely on the use of both adhesive patches (micropatterns), formed with adhesive proteins such as fibronectin or laminin, and repellent areas generated thanks to an anti-fouling coating. The quality of the anti-fouling coating – classically a polyethylene glycol (PEG) polymer – is critical to ensure the quality of the protein and cell micropatterning, in terms of longevity and shape accuracy. Indeed, when seeded, the cells should not be able to adhere to the repellent surface but only to the micropatterns.
Examples of micropatterning techniques include:
1. Microcontact printing
For this approach, a stamp with the desired microfeatures must be microfabricated using photolithography, photomasks and photoresists to generate a master displaying the designs of the future micropatterns. Polydimethylsiloxane (PDMS) is then poured on to the master and cured to create the stamp (opposite replicate of the initial master). The ‘ink’ for this stamp is a solution of adhesive protein. The non-patterned areas are then backfilled with an antifouling layer. This micropatterning technique is the one used most frequently and also considered the easiest (if buying the stamp from a provider instead of fabricating it in-house), but it is also the least precise one. Indeed, the sharpness of the stamp’s pillars edges, the pressure exerted on the stamp and the difference in protein quantity deposition, are all variable factors that ultimately affect the quality of the final micropatterns. And of course, at the microscopic scale, the alignment of the stamp on specific microstructures constitutes a real challenge.
2. Deep UV photolithography and photomasks
The advantages and drawbacks of deep UV photolithography through photomasks are directly correlated to its process. After coating the desired cell culture substrate with the anti-fouling polymer, a photomask, which is a robust and opaque stencil perforated with desired microscopic shapes, is placed over it. Deep UV light is then shined onto the top of the photomask to degrade the PEG layer on the areas matching the holes in the photomask. The solution-based adhesive protein is then added and adsorbs to the previously illuminated areas only. Here, the alignment issue and the possible damages caused to soft substrates by direct contact seem obvious. But when this technique is well performed it has the advantage of producing a large number of identical micropatterns. However, this efficiency in micropattern production is only valuable if the micropattern designs have been previously optimised for a specific experiment and cell type. Otherwise, a new photomask needs to be ordered, delaying the progress of the project.
3. Maskless and contactless photolithography
In 2016, the team of Dr Vincent Studer from University of Bordeaux, CNRS, presented a new micropatterning method based on the use of a digital micro-mirror device (DMD), also used in video-projectors, which they called light induced molecular adsorption of proteins (LIMAP) [8]. This technology has, since then, been improved and commercialised by Alvéole as the PRIMO device.
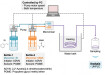
PRIMO is an optical module in which the main components are a DMD and a UV source (λ = 375 nm). This module can be installed on all the most common inverted microscopes. A dedicated software programme (named “Leonardo”) permits the selection, alignment, adjustment and replication of a preview of one or several desired micropatterns, of 1.2 µm minimum resolution, on the cell culture substrate placed on the motorised stage of the microscope. Once a micropatterning sequence is set, the software initiates PRIMO to project UV light onto the substrate previously coated with an anti-fouling layer and in the presence of a specific photo-initiator (PLPP). The combined action of the UV and PLPP locally degrades the anti-fouling coating, in only a few seconds. The protein is then added and adsorbs on the illuminated areas only, creating the micropatterns.
As a maskless and contactless system, the PRIMO system solves the issues of flexibility, alignment, substrate integrity and efficiency, which improves prototyping and optimisation capabilities. Indeed, the micropattern image can be projected on any standard cell culture substrate (stiff or soft, flat or micro structured, large or microscopic) and tested to get the best possible protein micropatterns for each specific experiment. Furthermore, as a photolithography technology PRIMO can also be used to fabricate 3D micro structured substrates (microfabrication and hydrogel polymerisation), combining structuration and functionalisation abilities in one unique tool.
Figure 3. PRIMO micropatterning platform. Credit: Alvéole.
Micropatterning: A fancy tool or a real asset for cell biology?
The recent progresses in bioengineering and bio-functionalised substrates opens up new opportunities to improve our understanding and questioning of cellular processes, extra-cellular matrix roles and related diseases [9]. Depending on the complexity of the scientific question, cell biology studies are conducted on different scales, going from single cells to small cell assemblies, tissues and, for a few years, organoids.
Even if micropatterning can be used for generating spheroids and analysing cell population forces, it is most applicable for studies on the cell cytoskeleton, traction forces, cell migration or disease modelling which often require single cell isolation or precisely controlled cell-cell interactions. Many experiments on cell forces and disease modelling are conducted on soft substrates. Here there are no doubts that the contactless micropatterning system PRIMO is a real advantage for this field as it is compatible with substrates of low stiffness; as shown by Dr Parameswaran’s team from Northeastern University in its work on airway smooth muscle cell contractions using micropatterning on gels of different stiffnesses ranging from 0.3 kPa to 40 kPa [10]. Another unique benefit of this innovative technology, not yet discussed, is the ability to locally control the protein density and generate gradients; making it a powerful tool for studying chemotaxis mechanisms or the immune response [11]. The growing number of research projects using this maskless, contactless technology confirms that researchers need to continue improving their control capabilities over the cell microenvironment in vitro by employing such a technology [12].
Cryo-electron tomography: The challenge of the perfect sample
In his 2012 paper published in Proceedings of the National Academy of Sciences (PNAS), Alexander Rigort stated “cryo-electron tomography provides unprecedented insights into the macromolecular and supramolecular organisation of cells in a close-to-living state”. Indeed, cryo-electron tomography has proven to be a great advance in microscopy to better visualise and understand intra and inter-cellular mechanisms in their native state and at the molecular level, such as viral infection mechanisms for example.
Figure 5. Tomogram from research on tethering proteasomes to nuclear pore complexes. Courtesy of Dr Ben Engel, Max Planck Institute of Biochemistry, Martinsried, for Leica Microsystems
But cryo-ET is still a young microscopy technique and all the steps of its workflow are being intensely reviewed and improved by both academic laboratories and industrial companies. Almost every step of the workflow is critical but in the case of eukaryotic cells, before even considering going to the microscope, the cell sample must meet some basic requirements: matching specific locations on the EM grid, containing the target component and being thin enough for transmission electron microscopy.
The ideal thickness for cryo-ET is estimated around 200 nm. As a comparison, depending on the cell types, the thickness of eukaryotic cells randomly spread on a flat surface can range between 0,5 µm (podosomes and lamellipodia) to 10 µm (centre of the cell around the nucleus).
By controlling the cell adhesion, spreading and shape, micropatterning overcomes the issues faced at the very first step of this workflow. Indeed, without micropatterning, cells in culture medium are randomly seeded onto EM grids and tend to adhere on the metallic mesh of the grid rather than the carbon film, preventing the electron to go through it when performing the transmission electron microscopy. Furthermore, the random seeding of cells is also correlated with a limited spreading making it more difficult to find the target due to the thickness. On the contrary, micropatterning allows the control over their location and the increase of cell spreading, therefore facilitating the next steps for cryo-ET cell sample preparation. However, microcontact printing and photopatterning with masks are very hard if not impossible to perform on EM grids due to their size and delicacy.
Several research teams have recently successfully micropatterned cells on EM grids using the contactless PRIMO micropatterning system and published their results showing unrivalled performances compared to other micropatterning techniques [13]. They were able to prepare better cell samples for cryo-ET experimentation, with cells well isolated and at the desired locations [14] and also emphasised that the impact of micropatterning on the organisation of the cytoskeleton could help predict the intra-cellular locations of some cytoskeleton components and improve the throughput of data acquisition, allowing for more efficient cryo-ET studies [15].
Integrating this new micropatterning method to the ongoing improvements in cell sample preparations for cryo-ET, including plunge freezing, correlative light microscopy, cryo-FIB lamella milling, as initiated with a partnership between Alvéole and Leica Microsystems, will soon accelerate research breakthroughs on what really goes on in the cellular machinery at the molecular scale.
References
1. M Bissell, M A Labarge. Context, tissue plasticity, and cancer: are tumor stem cells also regulated by the microenvironment?, Cancer Cell. 2005, 7(1):17-23
2. G C Reilly, A J Engler. Intrinsic extracellular matrix properties regulate stem cell differentiation. J Biomech, 2010, 43(1):55-62.
3. F M Watt, P W Jordan, C H O’Neill. Cell shape controls terminal differentiation of human epidermal keratinocytes. PNAS, 1988, 85(15):5576-80.
4. C S Chen, M Mrksich, S Huang, G M Whitesides, D E Ingber. Geometric control of cell life and death. Science, 1997, 276(5317):1425-8.
5. R McBeath, D M Pirone, C M Nelson, K Bhadriraju, C S Chen. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell, 2004, 6(4):483-95.
6. M Théry. Micropatterning as a tool to decipher cell morphogenesis and functions. J. Cell Sci., 2010, 123(Pt24):4201-13.
7. A Rigort et al. Focused ion beam micromachining of eukaryotic cells for cryoelectron tomography. PNAS, 2012, 109(12):4449-54.
8. P-O Strale, A Azioune, G Bugnicourt, Y Lecomte, M Chahid, V Studer. Multiprotein Printing by Light-Induced Molecular Adsorption. Adv. Mat., 2016, 28(10):2024-9.
9. V Ruprecht et al. How cells respond to environmental cues - insights from bio-functionalized substrates. J Cell Sci, 2017, 130(1):51-61.
10. S R Polio et al. Extracellular matrix stiffness regulates human airway smooth muscle contraction by altering the cell-cell coupling. 2019, 9(1):9564.
11. X Luo et al. Lymphocytes perform reverse adhesive haptotaxis mediated by LFA-1 integrins. J Cell Sci, 2020, 133(16):jcs242883.
12. Alvéole users’ scientific papers: https://www.alveolelab.com/resources-support-center/scientific-papers/
13. L Engel et al. Extracellular matrix micropatterning technology for whole cell cryogenic electron microscopy studies. J Micromech Microeng, 2019, 29(11):115018.
14. M Toro-Nahuelpan, L Zagoriy, F Senger, L Blanchoin, M Théry, J Mahamid. Tailoring cryo-electron microscopy grids by photo-micropatterning for in-cell structural studies. Nat Methods, 2020, 17(1):50-54.
15. L Engel et al. Lattice micropatterning of electron microscopy grids for improved cellular cryo-electron tomography throughput. BioRxiv, 2020
Digital Edition
Lab Asia 31.6 Dec 2024
December 2024
Chromatography Articles - Sustainable chromatography: Embracing software for greener methods Mass Spectrometry & Spectroscopy Articles - Solving industry challenges for phosphorus containi...
View all digital editions
Events
Jan 22 2025 Tokyo, Japan
Jan 22 2025 Birmingham, UK
Jan 25 2025 San Diego, CA, USA
Jan 27 2025 Dubai, UAE
Jan 29 2025 Tokyo, Japan