News & views
COVE Study reveals Phase 3 Results of Vaccine Selected by EC
Jan 05 2021
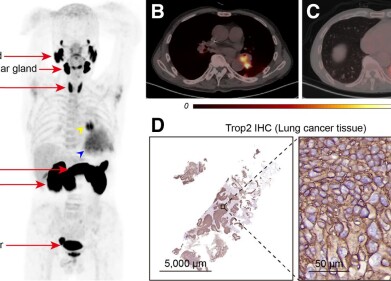
Results of the Phase 3 trial of Moderna’s COVID-19 Vaccine (mRNA-1273) released at the end of 2020(1) have indicated that the 100 μg two-dose regime of the company’s vaccine given 28 days apart was well-tolerated and demonstrated vaccine efficacy of 94.1% against COVID-19. The Phase 3 study, known as the COVE study, enrolled more than 30,000 participants aged over 18 in the US. It is being conducted in collaboration with the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH), and the Biomedical Advanced Research and Development Authority (BARDA), part of the Office of the Assistant Secretary for Preparedness and Response at the U.S. Department of Health and Human Services.
During November 2020, the European Commission approved an agreement to secure 80 million doses of Moderna’s mRNA-1273 vaccine candidate with the option to increase their purchase of mRNA-1273, from 80 million doses to a total of up to 160 million doses. Delivery of the vaccine could begin as early as the first quarter 2021 if it is approved for use by the European Medicines Agency (EMA) human medicines committee (CHMP), which started a rolling review of mRNA-1273 on November 17.
“We appreciate the confidence the European Commission has demonstrated in our mRNA vaccine platform by including mRNA-1273 in their portfolio of vaccines. We recognise that tackling this global pandemic will require a number of solutions and we are proud of the role Moderna has been able to play in this global effort,” said Stéphane Bancel, Chief Executive Officer of Moderna. “We have scaled up our manufacturing capacity outside of the United States with our strategic partners, Lonza and Rovi, to be able to deliver approximately 500 million doses per year and possibly up to 1 billion doses per year beginning in 2021, if approved.”
The Phase 3 COVE study is ongoing and will continue to follow participants for two years. Additional data to be collected will include longer term safety follow-up, duration of protection against COVID-19, and efficacy against asymptomatic SARS-CoV-2 infection. Moderna is also conducting a Phase 2/3 study of the Moderna COVID-19 vaccine in adolescents 12 to under 18 years of age. Additional studies are planned to evaluate the Moderna COVID-19 Vaccine in pregnant women, children younger than 12 years, and those in special risk groups, such as the immunocompromised.
(1) Published in the New England Journal of Medicine
Digital Edition
Lab Asia 31.6 Dec 2024
December 2024
Chromatography Articles - Sustainable chromatography: Embracing software for greener methods Mass Spectrometry & Spectroscopy Articles - Solving industry challenges for phosphorus containi...
View all digital editions
Events
Jan 22 2025 Tokyo, Japan
Jan 22 2025 Birmingham, UK
Jan 25 2025 San Diego, CA, USA
Jan 27 2025 Dubai, UAE
Jan 29 2025 Tokyo, Japan