-

-
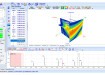
 Titration curve: Total hardness (Calcium and Magnesium hardness)
Titration curve: Total hardness (Calcium and Magnesium hardness)
Laboratory Products
Titration of amino hydro-chlorides (acc. to Ph. EUR): Application example for two equivalence points
Jun 06 2012
Up to now the amino hydro-chlorides were dissolved in glacial acetic acid, the amines released through the addition of mercuric acetate and titrated with perchloric acid in glacial acetic acid. According to the environmentally friendly method of the European Pharmacopeia the amino hydro-chlorides are dissolved in ethanol and being dosed with exact 5.00 ml of a 0.01 mol/l HCl. This mixture is then titrated with NaOH 0.1 mol/l.
Most titration curves show two equivalence points. The result is calculated from the difference between the first and second equivalence point. This method, with all parameters and calculation formulas, comes standard in the TitroLine® 7000 and can be used after the input of equivalent substance weight.
Learm more>>
Perfect for non-aqueous titrations
Eliminate the need for special electrodes (e.g. separate indicator, reference and auxiliary electrodes) with the built-in amplifier–ideal for titrations in non-aqueous solvents such as:
• Acid and base numbers in oils.
• Titrations in glacial acetic acid with perchloric acid.
• Hydroxyl, NCO (Isocyanate) number and further specific values.
pH Stat Titrations
With a pH stat application, a given pH is fi rst adjusted and then kept constant during the analysis with an acid or a base.
The pH stat titration is often applied to:
• Determination of the enzyme activity (ex. Lipase).
• pH stat elution of soil sample at pH 4.
• Monitoring of the pH value during chemical syntheses.
Digital Edition
International Labmate 49.6 - Sept 2024
September 2024
Chromatography Articles - HPLC gradient validation using non-invasive flowmeters Mass Spectrometry & Spectroscopy Articles - From R&D to QC, making NMR accessible for everyone: Putting NMR...
View all digital editions
Events
Sep 29 2024 Singapore
Oct 06 2024 Liverpool, UK
Oct 08 2024 Gothenburg, Sweden
Oct 09 2024 Birmingham, UK
Oct 09 2024 NEC, Birmingham, UK


.jpg)







-(300-x-250-mm)_1.jpg)




